Block 2 · More than just air: deco gases & helium
Which gas when – and why.
Introduction
This block is about gases that contain less nitrogen than air – and what that changes in terms of decompression, narcosis and planning. When does Nitrox make sense, where are its limits, and why is high-percentage Nitrox so efficient for decompression? At what point does helium become useful or even necessary – and what are the typical trade-offs?
This section does not replace practical training in technical diving. Its goal is to improve your understanding of decompression within this area of recreational diving, avoid common misconceptions, and make gas choices more transparent and logical.
Before we get started with Nitrox as a deco gas – have you already completed a standard Nitrox course? We won’t repeat that knowledge in full here. But you can first test your understanding and refresh it if needed before continuing.
Nitrox: oxygen sets the limits
Oxygen is essential for life – but at high partial pressures it can become toxic. For recreational divers, the most relevant risk is CNS toxicity: a seizure underwater is usually fatal. At the same time, it’s important to remember: within accepted limits, the risk is very low. We are working with deliberately conservative safety margins.
From your Nitrox course you’ll remember that paying attention to the MOD – the maximum operating depth –
is absolutely essential.
Exceeding 1.4 bar pO₂ during the bottom phase and 1.6 bar pO₂ during decompression should be avoided.
That may be most of what stuck.
But when it comes to deco gases, it’s worth revisiting oxygen in more detail.
Please refresh the concepts of MOD and best mix,
and take another look at CNS clock, CNS toxicity and pulmonary toxicity
before continuing with the text here.
SSI Nitrox course (online)
Oxygen exposure limits
Nitrox as a deco gas: why it is so efficient
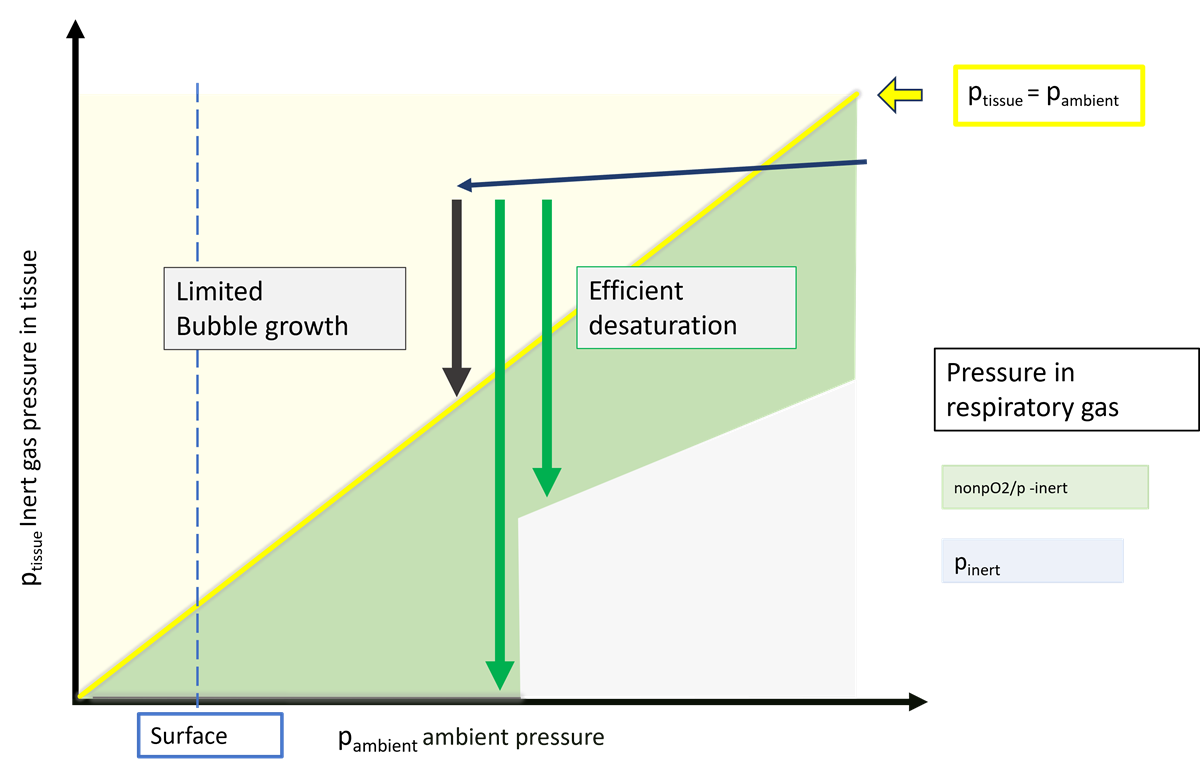
High-percentage Nitrox plays a very important role as a deco gas. This is not about “more oxygen = better”, but about less inert gas in the breathing gas, and therefore a stronger gradient that drives inert gas out of the body. Deco gases reduce the inert-gas partial pressure in the lungs, making off-gassing easier.
Using Nitrox as a deco gas has major advantages.
We’ve already seen, when discussing Gradient Factors, that a low GF Low enforces deeper stops.
At these deeper stops, medium and slow tissues continue to on-gas,
while only the fastest tissues are already off-gassing.
This creates a fundamental problem when choosing the first stop:
off-gassing becomes more efficient when the gradient between inert-gas pressure in the tissues
and inert-gas pressure in the lungs is higher.
With only one gas, the only way to increase that gradient is to ascend further.
At the same time, the risk of bubble formation increases with rising tissue supersaturation.
With a single gas, you have to accept that trade-off.
With a deco gas, you can decouple the two.
Reducing nitrogen in the breathing gas – and therefore in the lungs –
immediately increases the off-gassing gradient.
Medium tissues no longer continue to on-gas, or at least much less.
Yet you are still at a depth where bubble formation is less likely.
That is the key advantage of a deco gas:
it separates inert-gas gradient from ambient pressure.
In the fundamentals block you already encountered a very simple, didactic dive planner.
Here, deco gases now come into play.
You can plan deeper dives with up to two deco gases.
Before discussing which gases make sense for which dive,
take a look at a few profiles with different deco gases.
What changes during the ascent?
Pay attention not only to decompression time,
but also to which tissues start off-gassing after the gas switch
instead of continuing to on-gas.
Choosing the best deco gas
The planner is preloaded with two gases: EAN50 and pure oxygen.
This is a very common combination for deeper dives on air or normoxic trimix.
Together, they provide highly efficient decompression.
The first gas switch can already take place at 21 m,
and for dives typical in this range the gradient factor is still very low there.
If you only use a single deco gas, however, you have to decide what makes more sense:
EAN50, pure oxygen, or something in between.
You’ll have to make that decision yourself.
To gather the relevant information, try the following with the planner:
- Plan a dive on air to 40 m with a bottom time of 25 minutes, without a deco gas
- Use Gradient Factors that you consider reasonable
- Plan the same dive with EAN50 as a deco gas and compare the result
- Plan it again with O₂ as a deco gas – what changes now?
- Do you remember where the first stop was without a deco gas?
- Find the best mix at that stop depth for a pO₂ of 1.6
- Plan the dive again using that gas as a deco gas
- Then consider: which of the three options seems best to you?
In practice, you’ll often use whatever deco gas is available. Pure oxygen offers the most efficient off-gassing, but can only be used from 6 m. Depending on the dive profile, a gas with a slightly lower oxygen fraction may be the better choice.
Helium & trimix: why, when, and what for?
Helium is used to reduce narcosis and gas density. This can lower work of breathing and improve mental performance. At the same time, helium is significantly more expensive, and its specific properties must be considered during deco planning.
On deeper dives, using air as a breathing gas causes two problems
(which are closely linked physiologically):
nitrogen becomes narcotic with increasing depth,
and increased gas density makes breathing harder.
While “deep narcosis” itself is still not fully understood,
the physiology of increased work of breathing is well established.
As gas density rises, every breath requires more effort,
and maximum achievable ventilation decreases.
At extreme depths, this could even lead to a situation
where you simply cannot breathe enough.
Long before that, another effect may appear:
CO₂ levels in the body can rise.
Normally, elevated pCO₂ triggers increased breathing to eliminate it.
Underwater, however, some divers – so-called “CO₂ retainers” –
respond differently: their bodies tolerate higher CO₂ levels
to reduce the work of breathing.
Since CO₂ is highly narcotic,
this effect is suspected to contribute significantly
to the unpleasant effects often attributed to deep narcosis.
Limits for gas density and narcosis
What gas density is still acceptable,
and which mix is optimal?
Two factors matter when choosing the best mix
for a target maximum depth:
equivalent narcotic depth (END) and gas density.
To calculate END,
you determine at what depth air would have the same pN₂
as the trimix you intend to use at maximum depth.
Gas density at that depth is calculated
from the individual components oxygen, nitrogen and helium.
Decompression with trimix
Helium is much lighter than nitrogen and therefore diffuses into tissues much faster.
The Bühlmann model assumes that helium half-times are 2.65 times faster than nitrogen half-times.
Decompression therefore has to be calculated differently.
Beginners in trimix diving often assume that decompression should be shorter,
because the lighter gas leaves the body faster.
That assumption is wrong: decompression is usually longer.
The reason lies in deeper stops.
Because helium also enters tissues faster,
supersaturation during ascent is greater.
To off-gas fast tissues, stops have to begin earlier.
During those stops, medium and slow tissues continue to on-gas
and later have to off-gas at shallower stops.
With the planner you can observe tissue loading
in three heatmaps:
nitrogen only, helium only, and the combined inert-gas load.
At this point, you should already be using a different,
fully validated planner for real dive planning.
This tool is for visualisation only.
Still, you can explore different gases and their effect on runtime,
and better understand how helium fraction influences decompression.
How do you actually plan a trimix dive? You’ll find discussion of Gradient Factor choices in the block on current debates and research, and practical planning tips in the dive-planning block.